Most people who have eaten psychedelic mushrooms and then tried synthetic psilocybin, say that the mushroom hits different. For years, the scientific community largely treated this as anecdote.
But a study just published in Scientific Reports by Zurika Murray and colleagues at the University of the Free State in South Africa has offered what may be the most detailed mechanistic case for the mushroom “entourage effect” to date.
Beyond the single molecule
Most of modern pharmacology is built on the principle of isolating a single active compound, understanding its mechanism, and delivering it in a controlled dose. There is obvious wisdom in this. It’s how we got aspirin from willow bark and morphine from poppies. And it’s the logic that has driven the current wave of psychedelic medicine, with organisations like Compass Pathways and Usona Institute pursuing isolated synthetic psilocybin through FDA regulatory pathways.
But living systems rarely work through single molecules acting on single targets. Biology tends toward networks, redundancy, and context-dependence. A mushroom is not a pharmaceutical factory that happens to produce psilocybin. It is a living organism that has evolved a complex cocktail of bioactive compounds, each potentially modulating different aspects of mammalian neurochemistry.
The question Murray’s team set out to answer computationally is whether those other compounds actually do something meaningful once they reach the brain.
Eight compounds that cross the blood-brain barrier
The researchers began by cataloguing fifteen compounds known to occur naturally in Psilocybe mushrooms and then screening them for pharmacokinetic viability. Specifically, whether they could survive digestion, absorb through the gut, and cross the blood-brain barrier. That last criterion is the critical filter. A compound can have extraordinary receptor affinity, but if it cannot get into the brain, its neurological relevance is theoretical at best.
Eight compounds passed every test: psilocin (the active metabolite of psilocybin), norpsilocin, 4-hydroxytryptamine, 4-hydroxy-N,N,N-trimethyltryptamine, phenylethylamine, and three β-carbolines — harmane, harmol, and harmaline. All eight demonstrated high gastrointestinal absorption, blood-brain barrier permeability, and drug-likeness. All showed relatively low predicted toxicity, with oral LD₅₀ values generally exceeding 700 mg/kg.
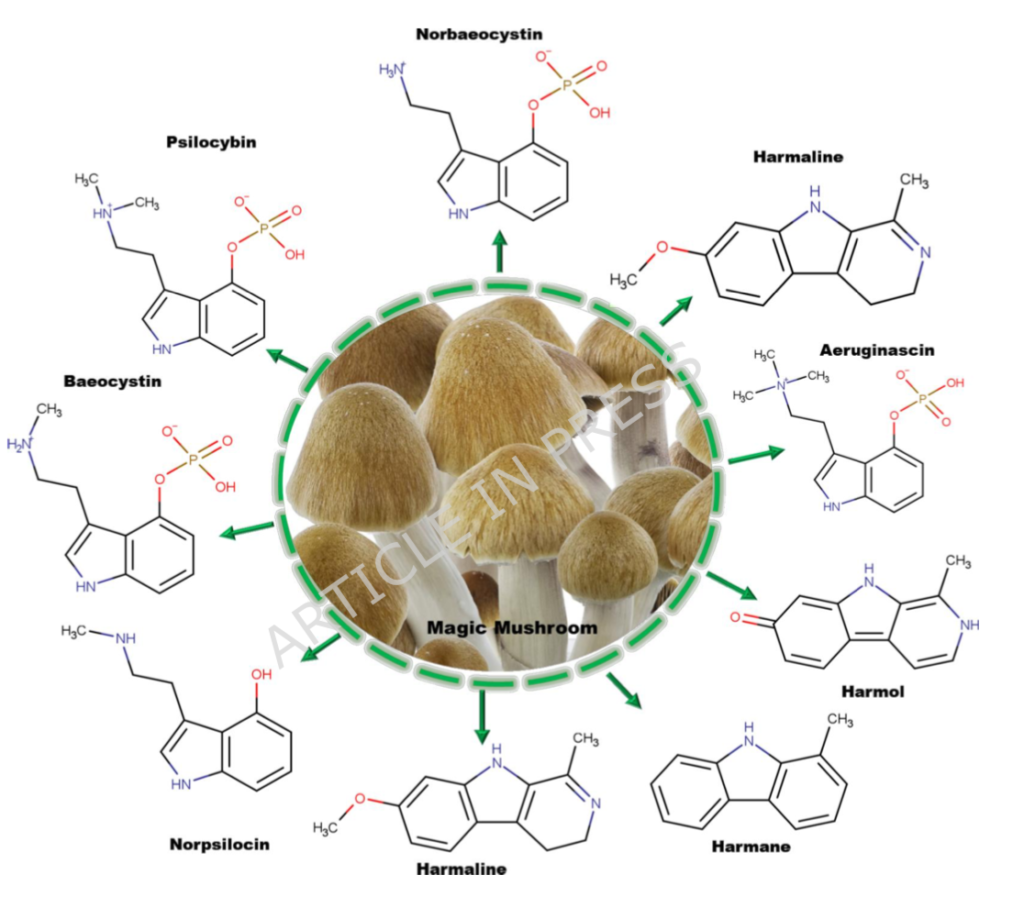
This alone is a meaningful finding. It establishes that psilocybin mushrooms deliver not one but eight brain-active compounds in a single dose.
The network is the point
Using a computational approach called network pharmacology, the team then predicted which human proteins these eight compounds might bind to. The result was a web of 44 unique protein targets, most of them expressed in the brain. When the researchers mapped the interactions between those proteins, they found something striking: the targets were not scattered randomly across unrelated pathways. They formed a densely interconnected network centred on serotonergic and dopaminergic neurotransmission – the systems that govern mood, cognition, perception, and reward.
The protein-protein interaction network showed statistically significant enrichment (p < 1.0e-16), meaning the connectivity between these targets was far greater than chance would predict. Two major functional clusters emerged. The first encompassed serotonin receptors (including HTR2A, HTR1A, HTR2C), dopamine receptors (DRD2), monoamine oxidases (MAOA, MAOB), and neurotransmitter transporters (SLC6A3, SLC6A4). The second cluster grouped adrenergic receptors involved in cardiovascular and CNS regulation.
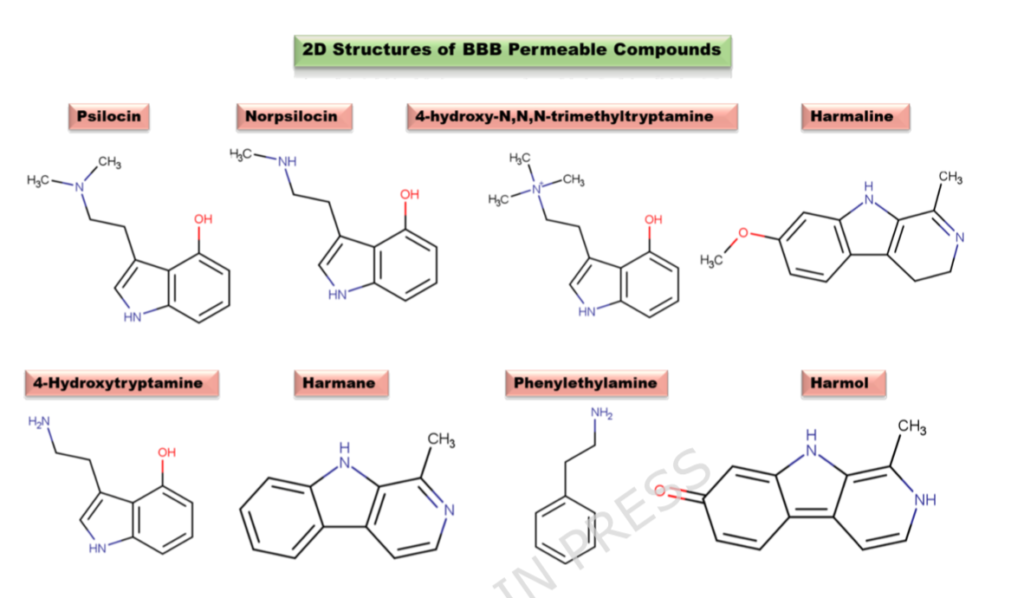
What matters here is the architecture of the interaction. These are not eight compounds each doing one thing. They are eight compounds engaging overlapping, functionally related targets across a coherent biological network. The enriched pathways read like a neuroscience textbook: serotonergic synapse, dopaminergic synapse, cAMP signalling, calcium signalling, and the cGMP-PKG pathway, which governs neuronal plasticity and cardiovascular response.
The ayahuasca parallel
Perhaps the most compelling finding concerns the β-carbolines harmane, harmol, and harmaline. The researchers demonstrated that these compounds bind stably to monoamine oxidase A (MAOA), an enzyme responsible for breaking down serotonin, dopamine, and norepinephrine in the brain. Harmaline showed an exceptionally low RMSD of just 0.09 Å within the MAOA binding pocket, indicating remarkable binding stability. Harmane and harmol displayed binding poses strikingly similar to harmine, a known reversible MAOA inhibitor.
If this sounds familiar, it should. Pairing a tryptamine with a monoamine oxidase inhibitor is precisely the biochemical trick that ayahuasca employs. In that traditional preparation, DMT provides the visionary tryptamine, while the β-carboline harmine (from Banisteriopsis caapi) inhibits MAO to prevent the DMT from being broken down before it can act. The mushroom, it turns out, may be running a gentler version of the same strategy, delivering both the tryptamine agonists and the compounds that slow their degradation, all in a single organism.
The implications are significant. By partially inhibiting MAOA, these β-carbolines could extend the half-life of psilocin and simultaneously elevate endogenous monoamine levels, potentially amplifying both the depth and duration of therapeutic effects. This may contribute to the longer-lasting benefits reported after whole mushroom consumption compared to synthetic psilocybin alone.
What the mouse data already hinted at
These computational findings gain additional weight from existing experimental evidence. A 2009 study by Matsushima and colleagues found that a crude Psilocybe argentipes extract was more effective than equivalent doses of pure psilocybin at reducing marble-burying behaviour in mice (a standard assay for anxiety) without affecting locomotion. More recently, Shahar et al. observed that whole psychedelic mushroom extracts produced different effects on synaptic protein levels compared to chemically synthesised psilocybin, hinting at distinct neuroplastic responses. And patient reports from clinical settings have consistently described a preference for mushroom-derived preparations in terms of experiential quality and therapeutic outcome.
Murray’s study does not prove the entourage effect in the way that a randomised controlled trial would. It is computational and predictive. But it provides the best mechanistic rationale yet for why the mushroom might outperform the molecule.
The regulatory landscape and the living whole
It is worth noting that the two US jurisdictions currently offering legal access to psilocybin (Oregon and Colorado) both use whole mushroom preparations in their regulated service programmes. The decision was partly practical, but it also reflects an intuition that the whole organism carries something the isolated compound does not.
Meanwhile, the pharmaceutical pathway necessarily pursues synthetic psilocybin, because regulatory frameworks are designed to evaluate single molecules with defined pharmacokinetics. Neither approach is wrong. They are addressing different questions under different constraints.
But if the mushroom truly is greater than the sum of its parts, then we are looking at a form of pharmacological intelligence that was not designed in a laboratory. It emerged through evolutionary pressures we do not fully understand, encoded in the secondary metabolism of a fungal organism that has been producing these compounds for millions of years.
What this invites us to reconsider
There is a broader pattern here that extends well beyond psychedelics. Modern science has been extraordinarily productive at taking complex biological systems apart and understanding individual components. But there is a growing recognition that the relationships between components often matter as much as the components themselves. A rainforest is not a collection of trees. A gut microbiome is not a list of species. And a mushroom, it increasingly appears, is not simply a delivery vehicle for psilocybin.
The entourage effect, if it holds up under further experimental scrutiny, would suggest that Psilocybe mushrooms represent something more like a naturally evolved pharmaceutical formulation.
This does not mean whole mushrooms are inherently superior for every clinical application. Standardisation, dosing precision, and reproducibility are important for rigorous medicine. But it does mean that in our urgency to isolate the “active ingredient,” we may have been overlooking the system it evolved within.
The magic in the mushroom is not just psilocybin. It’s the conversation between compounds. And that conversation, this research suggests, may be where the deeper therapeutic potential resides.
Source: Murray, Z., Lewies, A., Wentzel, J.F. et al. “Network pharmacology and molecular simulation reveal the entourage effect mechanisms of psilocybin-producing mushrooms on the brain.” Scientific Reports (2026). https://doi.org/10.1038/s41598-026-39483-7



Let’s consider the statement “Psilocybe mushrooms represent something more like a naturally evolved pharmaceutical formulation” scientifically. Evolution of such a complex formulation would require significant selection. Was this natural or human-guided selection? If natural, how does the effect of these mushrooms on mammals confer selective advantage?